BIOLOGY |
Comparisons in Physiology and Evolution, and Why Bees can Do the Things They Do |
Color vision
The Nobel prize winner Karl von Frisch (1886-1982) did not only unravel the mystery of bee dance communication. He also provided us with a window into the strange and altogether different world that bees see. For example, he discovered that bees can see polarized light. Even earlier, at a time when only humans were assumed to see in color, von Frisch discovered that bees can see colors. A few years later, his contemporary A. Kühn found that they can see ultraviolet light. Equally important perhaps, von Frisch and Kühn founded the Journal of Comparative Physiology in 1924 and so created a forum for a newly budding field. Through comparing the capacities of a wide range of animals, comparative physiologists hoped not only to discover ever more strange capacities in their subjects. They also hoped to develop a more and more complete picture of mechanisms realized in the animal kingdom. In both of these directions, through diligence and ingenuous experiments, comparative physiologists were extremely successful. They were sometimes less successful, however, in explaining the adaptive value of the mechanisms they had found, although this clearly was one of their goals. In fact, their explanations of why animals could do the things they do were often (but not always!) characterized by truly baroque speculations and pan-adaptionism. This was because some of them treated each species as an entity which had no evolutionary context or history. In a way, each species was thought to adapt individually to its particular environment, and its traits were freely variable in all directions. Other physiologists compared bees with humans (or other unrelated species), and the questions that arose from those comparisons seemed obvious: for example, why can insects (and other arthropods) see ultraviolet and polarized light when we can't? The answer appeared equally evident: there must be something in the environment of the animals that makes these feats advantageous.
But is this really so? One example will illustrate the problem. In a recent study, Brownell & Weber (1995) discovered that two species of strictly nocturnal species of scorpions were sensitive to polarized light. This appeared puzzling to Brownell & Weber, since the night sky does not contain any polarized light cues. But since there surely must be an adaptive explanation, they encouraged further exploration of the visual ecology of these nocturnal animals to find it. Unfortunately, polarization-sensitive receptors are not an adaptation in scorpions, or any other arthropods for that matter. Polarization sensitivity is simply an intrinsic property of the way their rhabdomeric photoreceptors are built. In fact, because such polarization sensitivity is often undesirable, many invertebrates have invented tricks to eliminate it (Wehner & Bernard 1993). These latter tricks are truly adaptations; polarization sensitivity in an arthropod photoreceptor is not. The answer to the question why nocturnal scorpions see polarized light is simply this: because their ancestors did!
How can we infer how non-extant species saw the world? Since time machine projects habitually run into complications, it is now difficult to obtain funding for them. Therefore, evolutionary biologists resort to an alternative strategy called the comparative method (Harvey and Pagel 1991). Such an analysis is based on the observation that biological traits are more likely to stay the same than to change; thus, if two sister taxa share a common trait, we assume that it is more likely that this trait evolved once in the ancestor of the two taxa, rather than twice, independently, in both of them. Therefore, because all arthropods see polarized light, nocturnal scorpions did not evolve this capacity; they had it from the start, and there was no selective pressure to get rid of it. In such a case, it is futile to search for an adaptive explanation. It is this simple evolutionary reasoning that is missing in many physiological studies. Strangely, however, some workers have flatly denied that physiologists should look at phylogenetic trees: "...comparative physiology needs the collaboration of physiological ecology more strongly than that of evolutionary history or systematics. In fact ... comparative ... physiology seems mainly dependent ... on adaptive explanations..." (Waterman 1975). As the above example shows, this claim is hardly tenable. Comparative physiology needs systematics to understand adaptation. We will support this claim with two examples of von Frisch's favorite topics - bee color vision and bee dances.
The discoveries that bees see ultraviolet and that flowers reflect such short wavelengths were made in the 20s (see references in von Frisch 1965). Ever since that time, scientists have speculated that UV receptors in bees had developed in a co-evolutionary process with floral coloration. This notion was given recent impetus when it was found through computer modeling that bee color vision is indeed the optimal color vision system to detect and identify flowers (Chittka 1996a). But does the evolutionary influence between bees and flowers really go in both directions? To prove that flower signals indeed drove the evolution of bee color vision, it must be shown that the ancestors of bees possessed different sets of color receptors prior to the advent of the flowering plants. But how can we determine in what colors insects saw the world 200 million years ago? One has to evaluate members of Arthropod taxa whose evolutionary lineages diverged from those of bees before there were flowers. If the color vision of such animals is indistinguishable from that of bees, this implies that it was present in an ancestor of bees which predated the evolution of flower color. Fortunately, comparative physiologists have collected an extremely large data base on color receptors in various arthropod taxa (e.g. Menzel et al. 1986). We only need to map them on the phylogenetic tree, and any patterns of adaptation should be immediately apparent.
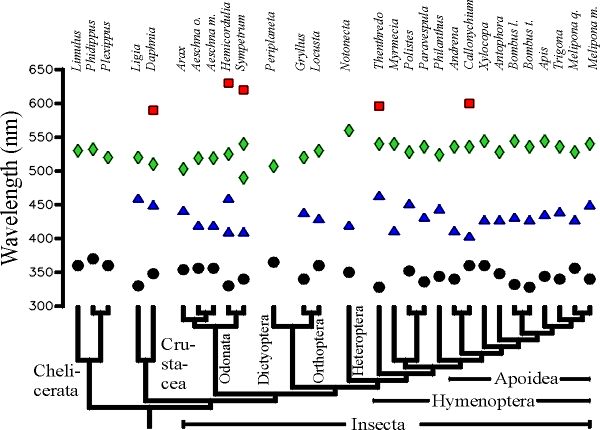
Fig. 1 The maximum wavelength values of photoreceptors of 29 species of Arthropods superimposed on the phylogenetic tree of these species. O - UV receptors; D - blue receptors; à - green receptors; o - red receptors. For complete species names and original data the reader is referred to the literature cited in Chittka (1996a).]
The lmax values of the Crustacea and Insecta fall into 3 distinct clusters around 350, 440 and 520nm (Fig.1). Most Chelicerata lack blue receptors. In contrast, almost all Mandibulata (Crustacea and Insecta) possess at least the three color receptor types maximally sensitive in the UV, blue, and green (Chittka 1996b). Thus, the blue receptor appears to be an evolutionary novelty in the ancestor of the Mandibulata. The few insect species in which one of the three types is absent (Periplaneta and Myrmecia) clearly represent cases in which these receptors were lost secondarily. Red receptors show up irregularly both in the Crustacea and Insecta; they have evolved several times independently. The photoreceptor wavelength positions of UV-, blue and green receptors are surprisingly conserved in the Mandibulata.
Thus, we can infer that the Cambrian ancestors of extant insects and crustaceans possessed UV-, blue- and green receptors, while the common ancestor of the Mandibulates and Chelicerates lacked blue receptors. Insects were well pre-adapted for flower color coding more than 500 million years ago, about 400 million years before the extensive radiation of the flowering plants which started in the middle Cretaceous (100 million years ago), although their origins might have to be placed in the Triassic. Recent data on the molecular structure of photopigments support the interpretation that the basic types of Arthropod visual pigments must be placed at the very roots of Arthropod evolution (A. Briscoe, personal communication). Why are the wavelength positions of color receptors so conservative in many arthropods? Generating photopigments with lmax in certain regions of the wavelength range between 320 and 630nm does not constitute a technical impossibility, since there are at least a few species in the Crustacea and the Insecta whose retinae contain 5 or more visual pigments, some of which fill the "empty spaces" on the wavelength scale commonly not occupied by arthropod lmax values. A more likely explanation is that genetic variability to modify peak absorbency of pigments has not existed in the species in question (Goldsmith 1990). Indeed, molecular studies on insect visual pigments show that mutations at several sites are necessary to cause a modification of the spectral sensitivity of a photoreceptor (Britt et al. 1993).
In summary, the answer to the question of why bees possess UV-receptors is the same one as above: because their ancestors did! The widespread set of UV, blue, and green receptors in Arthropods might actually be adaptive, but to an unrecognized set of environmental parameters. The hypothesis that insect color vision was adapted to particular classes of objects, such as flowers in the case of bees, can be rejected. At best, UV-blue-green trichromacy might constitute a more general adaptation which provides at least a local optimum for coding all sorts of natural objects under various illumination conditions (Chittka 1996a).
Bee dances
Von Frisch's analysis of the bee dance language remains the most fascinating discovery in non-human communication. Besides humans, only bees appear to rely on a symbolic language to tell conspecifics about the location of a profitable food source. Successful foragers return to the hive, and perform a series of rigorously stereotyped motor patterns, roughly in figure-eight design. The speed of these dances codes distance, whereas its alignment with gravity codes the direction of the food source relative to the sun. The reader is referred to von Frisch's (1965) excellent monograph for details of these "bee dances". How did this fascinating behavior evolve? Surprisingly, there have been attempts to understand the evolution of bee dances entirely without any phylogenetic reasoning (Gould & Towne 1986). Unsurprisingly, however, such attempts have remained unsuccessful. Equally unsuccessful were attempts to understand the evolution of bee language by describing remotely similar behaviors in some solitary insects such as flies or moths (von Frisch 1965). Interesting as they are, such observations cannot be used in an evolutionary analysis, because the species are too distantly related. Von Frisch's most successful student, Martin Lindauer, rightfully thought that the answer can only be found by looking at closely related species of our honeybees (reviewed in von Frisch 1965). Lindauer's reconstruction of the evolution of bee dances is an early masterpiece of tracing the evolution of complex behavioral traits. He examined three species of tropical honeybees and a variety of the honeybees' closest relatives, the stingless bees. We only briefly allude to these results here because they have been described elsewhere (Lindauer & Kerr 1958, von Frisch 1965).
The picture that emerged from Lindauer's studies is this: dances of all species of the genus Apis consist of similar motor patterns, and the dances all contain information about distance and direction of a profitable food source. In all species, direction to a food source is assessed as the direction of the food source relative to the current position of the sun. In all species except the one considered most "primitive" (Apis florea) this angle is expressed during the dance (which is naturally performed on a vertical surface) as the angle relative to gravity. A. florea, which nests in the open, and whose dancers dance on a horizontal surface, does not perform the transformation relative to gravity: its foragers orient their dances relative to the direction of the sun, as it is seen on the flight to the feeder. Lindauer thought this to be the original form of the dance. This is not necessarily true. It is a widespread assumption of many non-evolutionary biologists that extant species which diverged earlier from a common tree (the more "primitive" species) might have somehow conserved earlier forms of a behavior or other traits, and so, in the case of bees, to provide a direct possibility to observe the ancestral forms of the dances which von Frisch observed in the honeybee Apis mellifera. But the ancestral species had as much time from the point of divergence as the derived species (that arose from the sister species of the ancestral species) to evolve communication systems, and even in species whose morphology and nesting habits have remained largely conserved since the time of divergence, behavior might have undergone adaptive change. Reconstruction of the ancestral dance is only possible through identifying dance features that are shared between sister taxa, and identifying outgroups which lack these traits. In a more recent phylogenetic reconstruction of dance evolution within the genus Apis, Dyer (1991) points out that it is equally parsimonious to assume that A. florea secondarily lost the ability for the transformation.
Do stingless bees, the sister group of the honeybees, contain information about which scenario is correct? Unfortunately not. None of their numerous species seem to perform the kinds of highly repetitive motor patterns called dances in the honeybee. Most, however, do perform excited (or excitatory?) runs on the comb once they return from a successful foraging bout (Lindauer & Kerr 1958). Several species also emit small vibrational pulses during these runs, which are correlated in length with the distance of a food source, as in honeybees (Esch 1967). With these they also share the use of trophallaxis: successful foragers often regurgitate nectar during their dances (or excited runs, if you prefer not to call the chaotic motor patterns of stingless bees "dances"). Besides these shared traits, stingless bees show strong differences in recruitment systems between species. Some use scent trails, and some guide nestmates directly to a food source. In conclusion, the common ancestor of the stingless bees and honeybees likely did perform excited runs on the comb after finding food, did have trophallaxis, and did use sound to alert nestmates and possibly to code distance. How the ancestor of the honeybees evolved the more elaborate form of the dance - whose discovery won von Frisch the Nobel prize - we will probably never know. There is simply too large a gap between the behavior of the honeybees and that of their closest relatives. Some workers have suggested that the direction code of the honeybee dance might have evolved from "intention movements" as found in some stingless bee species (von Frisch 1965, Esch 1967). These stingless bees of the genus Melipona make repeated "false starts" in the direction of the food, but return back only to take off another time. But even though Melipona is considered an "advanced genus" of stingless bee, it is not the closest relative of the honeybees! Instead, the closest relative of the honeybees' ancestor is the ancestor of all stingless bees. Since false starts are not universal in the stingless bees, they are most likely not the ancestral condition; if they are not the ancestral condition in stingless bees, we cannot claim that they are ancestral in the sister group of the stingless bees, the honeybees. Thus the origin of honeybee dances, including their symbolic code of distance and direction, is likely to remain an enigma.
But the very origins of the "dances" of the Cretaceous ancestor of the eusocial Apid bees (honeybees, stingless bees, and bumble bees) are still in the dark as well. How did dancers and recruits first establish contact? Which evolved first, the peculiar behavior of successful foragers to run around excitedly in the nest, or the readiness of potential recruits to follow successful foragers? Clearly, both need to exist for recruitment to work. But why would any bees follow a successful forager if such a forager has not yet "invented" a message that, at the very least, identifies it as a successful forager? And surely, it is just as hard to conceive that successful foragers perform excited motor patterns in the nest if no one attends (because bees of the pre-dance time had no yet "invented" their dance-following behavior). Yet, von Frisch (1965) cites evidence that bumble bees, the closest relatives of the stingless bees and honeybees together, do exactly that. Earlier workers had observed that bumble bees returning from a feeder will run around in a seemingly excited manner on the nest, and occasionally bump into nestmates (much as several species of stingless bees will) - but no one cared about these maneuvers - other bees seemed not at all impressed by such excited foragers and could not be recruited to a food source. We examined three species of bumble bees (Bombus impatiens, B. occidentalis, and B. terrestris) and observed similar behaviors as were previously described: a returning successful bee will not just unload her crop, but perform extended runs across the combs, up and down the walls of the nest, and will sometimes even leave the nest only to turn back an perform more zigzag runs on the combs. This behavior may last several minutes before the forager heads back to the feeder. But none of the bees in the nest ever followed the "dancing" bee. So could this mean that "dancing" really preceded the evolution of dance-following? Did dancers start sending messages before anyone "heard" them? If colony fitness depends on food intake, why would foragers waste precious time that could be spent collecting food? If there was no immediate fitness advantage of such strange behavior, how could it evolve?
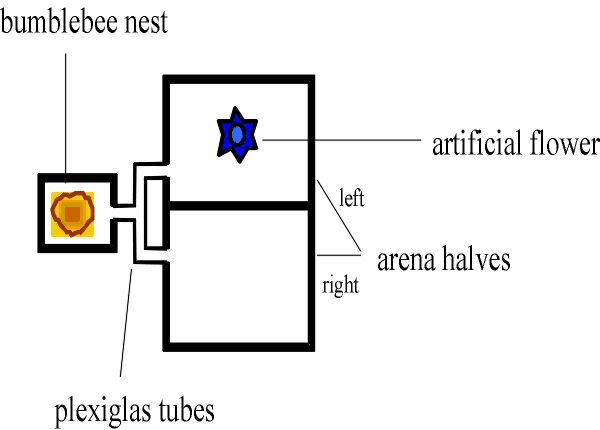
Fig. 2. Setup for testing if bumble bees show recruitment. A single bee continuously shuttled back and forth between the nest and a feeder in the left half of the arena, to collect as much nectar as possible. To see if she transmitted information about the existence of the feeder was transmitted in the nest (rather than at the feeder itself) we deviated all recruits into the right half of the arena.
We suspected that there must be a message, after all. To test this possibility, we allowed only a single bumble bee to collect nectar at a feeder in a flight arena (an experimental box of 40cm´ 60cm with a transparent cover to observe a bee's behavior). Once the bee discovered the nectar, she continuously shuttled between the feeder and the nest to bring more and more stomachloads full of sweet goods home. During its stays in the nest, we observed its interactions with nestmates (Dornhaus et al. 1999). We found that, although other bees did not follow the rewarded bees' "dances", they were very interested in the food she brought home. Immediately after she unloaded into one of the nest’s numerous honeypots, other bees probed the new nectar. How did they know that new nectar had been brought in? Somehow the returning forager must have alerted the other bees. But even more surprisingly, we found that if that single forager had brought several foraging loads home, the entire forager force of the colony would leave the nest and search outside for food! We explicitly made sure that the rewarded bee could not recruit the other bees by scent-marking the feeder. This was done by deviating the recruited bees into a different arena. So the primitive dances were not senseless - they did serve to motivate other bees to seek food! But how was the message transmitted, if not through mechanical contact? One possibility might be that bees waiting in the nest simply reacted to a sudden increase in nectar supplies, and took this as indication that rich sources of food had been discovered. Indeed, we were able to mimic the effect of a regularly returning successful forager by introducing a "ghost forager": we regularly introduced small quantities of nectar into the colony's honeypots by means of a syringe, and chose the amounts so that they approximately matched those of a real forager bee. This ghost bee motivated many bees to leave the nest and search - but not quite as many bees as a real bee. There must have been an additional signal transmitted while the rewarded bee was running around the nest. To test what this signal might be, we placed two nests right next to one another, but separated them by means of two layers of wire mesh, far enough apart that bees from the two colonies could not directly contact each other. Next, we rewarded bees of one nest by offering food in a flight arena, and we counted bees entering another arena that was connected to the second nest. Indeed, the bees in the unrewarded nest left their home in search for food when the neighbor colony was rewarded! This could mean that successful bees emit a pheromone that informs other bees of the existence of valuable food sources. It is also possible that bees emit substrate vibrations which alert other foragers, as do honeybees and stingless bees. In fact, bumble bee foragers do emit characteristic short buzzes when returning to the comb, but these buzzes are independent of whether the returning bee is a successful or unsuccessful forager, and independent of how far it has traveled. The sounds are even produced when a bee has only briefly left the comb, and not even flown to forage (Heidelbach et al. 1998, H. Böhm, personal communication). When we separated the two nests by clear plastic wrap, so maintaining visual contact between the two colonies, and the possibility of vibration transmission, we found that the unrewarded nest stopped responding. Therefore, the excited runs by bumble bee foragers (and possibly the ancestor of all eusocial bees) likely served the purpose of distributing a pheromone "message" as widely across the colony as possible. Admittedly, this scenario is speculative, but at present there is no better explanation for why foragers which have discovered a rich food source should invest any time into performing however articulate motor patterns if these are not paid attention to. Obviously, our scenario would gain much more weight if we could show that such pheromones are emitted during dancing in other social bees, such as stingless bees and honeybees. There is preliminary evidence to show that, in honeybees, this is indeed the case (J. Tautz, personal communication). Clearly, we need data from stingless bees as well. When these data are at hand, we are confident that it should soon be possible to reconstruct the early origins of bee communication!
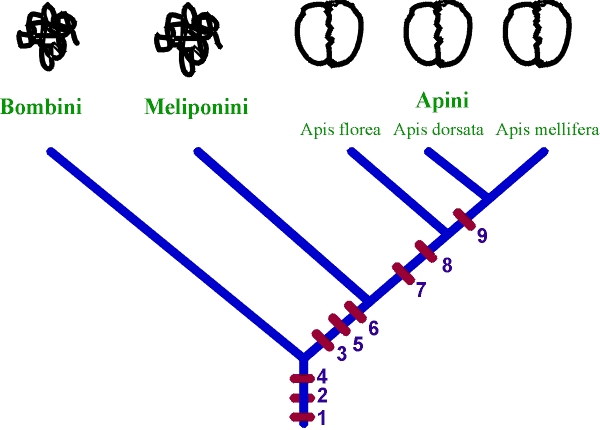
Fig. 3 Major events in the evolution of bee dances, mapped on the phylogenetic tree of the eusocial bees. Basic structures of "dances" in various taxa are shown on top. 1. excited runs on the nest by succesful foragers; 2. workers probe nectar that has been brought into the nest; 3. sound/vibration pulses produced by returning foragers; 4. trophallaxis; 5. length of buzzes correlates with distance to food; 6. "dance"-following, or at least "turning responses" towards the successful forager; 7. figure-eight waggle dance pattern; 8. dance performed on horizontal surface; 9. waggle dance performed on vertical combs. An additional event at the very root of the tree may have been the spread of a pheromone which is distributed by successful foragers.
We hope to have convinced the reader that comparisons between sister taxa are necessary to trace the evolution of bee communication, and for that, we obviously need systematics. Clearly, then, our interpretations depend on the particular phylogenetic tree we use as a basis. If it turns out that bumble bees, not stingless bees, are the closest relatives of honeybees, this will change our understanding of bee recruitment systems considerably. But at the moment, it appears that the phylogeny is as outlined above (Roig-Alsina & Michener 1993). Hopefully, it has also become clear that we cannot understand adaptation by looking at species as isolated entities which can freely vary their behavioral traits to match any environmental conditions. For example, the dance language of the honeybee Apis mellifera has been considered particularly efficient for exploiting the floral markets of temperate habitats (von Frisch 1965). Even if that should be true (which is difficult to test!), this clearly does not mean that the dance language evolved in Apis mellifera as an adaptation to communicate floral offerings in temperate climate. The ancestor of all extant honeybees likely lived in the tropics, and the dance language evolved in that ancestor (Dyer 1991). Therefore, it is not an adaptation to anything temperate (although it is unlikely to be of disadvantage in such habitats). But is the dance language an adaptation to particularly patchy floral food sources in tropical habitats, in which arboreal nectar sources are more common? In such conditions recruits can be allocated rapidly to a newly blooming tree, before it is discovered and depleted by other foragers. Unfortunately, stingless bees also live in tropical forests, and they do not use the same language. Some species may have not (yet) "invented" it, but there are others that likely never will: some species of stingless bees are more efficient in recruiting nestmates by using scent trails, and scouts escorting recruits, than are honeybees using their more elaborate dances (Lindauer & Kerr 1958). In such species, a mutant that "introduces" honeybee dances might be selected against, because it might make social foraging less efficient (unless the costs of scent trails greatly outweigh the costs of dancing).
In conclusion, dances we know from honeybees are clearly not the only possible result of adaptive evolution of bee communication in tropical habitats. The take home message is this: different animal species often live in conditions which differ along many dimensions. They also often differ in many traits, be they morphological, behavioral or physiological. For any unique trait in any animal, it is generally quite easy to find some unique environmental conditions to which that trait could have adapted. Without very much caution, interpretations of such correlations are bound to remain speculative. And clearly, we need to take into account a species' history to understand adaptation.
Finally, another reason why we know so little about how many physiological or behavioral traits evolved is the disregard of many physiologists for inter-individual variance. They often treated such variance as noise, which needed to be eliminated by averaging large numbers of measurements from different animals. And animals which did not display the supposed behavior sometimes never even entered data analysis. When we discussed this problem with M. Lindauer, he recounted the following anecdote. In 1952, he worked with von Frisch on the question of whether honey bees can communicate height in their dances. To this end, they trained bees from a hive at the bottom of a valley to a feeder on a bridge 76m high. Von Frisch (monitoring dances at the hive) and Lindauer (who guarded the feeder on the bridge) communicated by means of a tin hanging from a rope, which contained little notes on paper. On one occasion, von Frisch demanded, through such a note, the "EXECUTION OF THE FOLLOWING BAD DANCERS: ...!"; this was followed by a list of the doomed bees carrying the unfortunate labels. This is more than an anecdote about data treatment in the 50s! For many decades, scientists have lumped responses from many animals, and eliminated responses which showed gross deviations from the expectation. Sometimes this may be legitimate. Behavioral data and physiological measurements are often so noisy that extracting any information at all is not possible without averaging. And strong deviations from expected observation will in fact often mean that the measurement is imperfect, for example in electrophysiology. But we may have lost much valuable information through such averaging! To return to an example above, could it be that the reason for much of the conservatism in arthropod color receptors exists because there simply is no variance between individuals of the species in question? A large number of scientists has worked on, e.g., the color receptors of honeybees, and the results differed within studies as well as across studies. But the debate about these differences mostly focused on the possible contributions of artifacts or different electrophysiological methods (Menzel et al. 1986). To be sure, both of these may add noise to the measurements, but unfortunately, the possibility that interindividual variance may also contribute has not even been considered. Therefore, we do not know if bee color vision systems even contain the raw material for evolution.
J. Gould wrote about von Frisch: "His pioneering work inspired the discovery of several otherwise unimaginable sensory systems in animals: infrared detectors in night-hunting snakes, ultrasonic sonar in dolphins and bats, infrasonic hearing in birds, and magnetic field sensitivity in a variety of animals. Doubtless, other systems are still to be discovered. The lesson is a melancholy one: We are blind to our own blindness, and must try not to read our own disabilities into the rest of the animal kingdom." (Gould 1980). We could not agree more strongly! If, in addition, we try not to be blind to recent (and not so recent) developments in evolutionary biology, we may soon be able to explain why we are blind to some things that animals can perceive, and why the rest of the animal kingdom lacks some capacities we pride ourselves upon.
References
Bibliogrphy
Britt, S. G., Feiler, R., Kirschfeld, K. & Zuker, C. S. (1993). Spectral tuning of Rhodopsin and Metarhodopsin in vivo. Neuron, 11: 29-39.
Brownell, P. & Weber, T. (1995). Polarization vision in nocturnal sand scorpions. In: Nervous Systems and Behavior. Eds: Burrows, M., Matheson, T., Newland, P.L. & Schuppe, H. Thieme Verlag, Stuttgart, p. 281.
Chittka, L. (1996a). Optimal sets of colour receptors and opponent processes for coding of natural objects in insect vision. J. Theor. Biol., 181: 179-196.
Chittka, L. (1996b). Does bee color vision predate the evolution of flower color? Naturwissenschaft, 83: 136-138.
Dornhaus, A., Hartmann, F. & Chittka, L. (1999). A window into the past: what bumble bees tell us about Cretaceous dances. In: Goettingen Neurobiology Report 1999, Eds: Elsner N. & Eysel U., Thieme Verlag, p.557.
Dyer, F. C. (1991). Comparative studies of dance communication: analysis of phylogeny and function. In: Diversity in the genus Apis. Ed: Smith, D.R., Westview Press, Boulder, pp. 177-198.
Esch, H. (1967). The evolution of the bee language. Scientific American, 216: 96-104
Frisch, K. v. (1965). Tanzsprache und Orientierung der Bienen. Heidelberg: Springer.
Goldsmith, T.H. (1990). Optimization, constraint, and history in the evolution of eyes. Quart. Rev. Biol., 65: 281-322.
Gould, J.L. (1980). Navigation by honey bees. In: Genes, Cells, and Behavior. Ed: N. Horowitz & E. Hutchings, W.H. Freeman, San Francisco, pp. 135-141.
Gould, J. L. & Towne, W. F. (1986). Evolution of the dance language. American Naturalist, 130: 317-338.
Harvey, P.H. & Pagel, M.D. (1991). The comparative method in evolutionary biology. Oxford University Press
Heidelbach, J., Böhm, H. & Kirchner, W.H. (1998). Sound and vibrational signals in a bumblebee colony (Bombus terrestris). Zoology: 101: 82.
Lindauer, M. & Kerr, W. (1958). Die gegenseitige Verständigung bei den stachellosen Bienen. Z. Vergl. Physiol., 41: 405-434.
Menzel, R., Ventura, D. F., Hertel, H., de Souza, J. M. & Greggers, U. (1986). Spectral sensitivity of photoreceptors in insect compound eyes: comparison of species and methods. J. Comp. Physiol. A, 158: 165-177.
Roig-Alsina, A. & Michener, C. D. (1993). Studies of the phylogeny and classification of Long-tongued bees (Hymenoptera: Apoidea). Univ. Kans. Sci. Bull. 55: 124-162.
Waterman, T.H. (1975). Expectation and Achievement in Comparative Physiology. J. Exp. Zool. 194: 309-344.
Wehner, R.& Bernard, G. D. (1993). Photoreceptor twist: A solution to the false-color problem. Proc. Nat. Acad. Sci. (USA), 90: 4132-4135.
Pointers of Interest
1. Schematic movie of a bee dance
http://ourworld.compuserve.com/homepages/Beekeeping/beedance.htm
2. The bee dance deciphered
http://www.bhs.mq.edu.au/psy/105/lectures/chris2/Week13_p1.html
3. How bees see the world
http://cvs.anu.edu.au/andy/beye/beyehome.html
4. How bees see flowers
http://gears.tucson.ars.ag.gov/ic/vision/bee-vision.html
5. Information about bumble bees
http://www.pollination.com/IPSbumbee.html
Glossary
Polarized light: Light has wave characteristics, and if these waves are aligned so that they all oscillate in a particular direction, light is polarized. Normal skylight, for example, contains strong components of polarised light, and the patterns of skylight polarisation allows bees to calculate a compass direction.
Pan-adaptionism: A philosophy by which all traits observed in present species are inherently optimal and advantageous for their bearers. But this is not always the case! For example, all insects have six legs. This does not mean that six is the optimum number of legs to cope with any task an insect ever faces - but they have to somehow deal with the equipment they have inherited! Individual insect species have no possibility to vary this number. In this case, the trait (having six legs) reflects a phylogenetic constraint rather than an adaptation.
Phylogeny: Phylogeny is the science of reconstructing the degree of relatedness between different animal species.
Phylogenetic tree: a phylogenetic tree is a tree which maps the relationship of animal species. Speciation takes place by seperation of one species into two species; therefore, all tree branchings are bifurcate. Species that are closely related (and which diverged in comparatively recent evolutionary time) are located closer together on the tree, while unrelated species (whose lineages have diverged a long time ago) are far apart in the tree.
Arthropods: A large group of animals which contains more than 75% of all know species. They are characterised by an external skeleton, and extremities consisting of several movable parts. The Arthropods comprise animals such as the spiders, Crustaceans and Insects, and the extinct Trilobites.
Photoreceptors: are color-sensitive cells in the eyes which send electrical signals to the brain. Each photoreceptor is particularly sensitive at one particular wavelength (its lmax). For example, UV receptors respond most strongly to UV light, whereas blue receptors are most sensitive to blue. But photoreceptors are somewhat sensitive at neighboring wavelengths as well. In fact, sensitivity falls to both sides from the lmax in the shape of a bell-shaped curve. Even though most animals have only a few classes of photoreceptors (blue, green, and red in the case of humans; UV, blue and green in the case of bees), the brain can use the ratios of signals from these few photoreceptors to perceive thousands of different colors.
Rhabdomeric photoreceptors - elongated photoreceptor cells which have small tubular extensions which usually point all in one direction, so that the rhabdomeric photoreceptors look roughly like a toothbrush. These tubular extensions - the microvilli - contain the photopigment molecules. The structure of the microvilli forces the photopigment molecules to be aligned all in one direction — the longitudinal direction of the microvilli. This alignment causes the polarisation sensitivity: each photopigment molecule is a dipol and is thus particularly sensitive to light that oscillates in one direction. Because the photopigment molecules are all arranged pointing in the same direction in the rhabdomeric receptor, such receptors are maximally stimulated by light oscillating in the same direction as the longitudinal direction of the pigment dipols. All insects and Crustaceans have such rhabdomeric receptors. Conversely, humans do not have such receptors — their photoreceptors contain randomly arranged photopigment molecules. Therefore, humans are insensitive to polarised light.
Taxa / sister taxa: A taxon is a group/category of organisms/species that is clearly distinguishable from another such group. The group must share a common ancestor; it must be monophyletic. A species is a taxon, but also a group of species that has a common ancestor, such as the insects or the mammals. Some taxa were previously regarded as having a single common ancestor, but closer inspection has revealed that they have several unconnected roots (e.g. Funghi). Such categories no longer have taxon status. Two groups of animal species which share a single common ancestor which is unique to them, are called sister taxa.
Selective pressure: environmental pressure that favors individuals with certain genes over individuals with other genes.
Lineages: is a continuous unidirectional path along a phylogenetic tree.
lmax or maximum wavelength: see photoreceptors.
Crustacea: primarily aquatic animals with notably strong armour, including e.g. lobsters and crabs.
Insecta: typically have 6 legs and a body composed of three conspicuous parts. Insects comprise such animals as locusts, bees, butterflies, beetles, flies, cockroaches and dragonflies.
Chelicerata: A group of animals which contains the spiders, horseshoe crabs, and scorpions
Mandibulata: a collective term for the Crustaceans and insects together.
Cambrian: Geological period 590-505 million years ago
Cretaceous: Geological period 130-65 million years ago
Triassic: Geological period 250-210 million years ago
Photopigments: are light-sensitive proteins in the membranes of photoreceptor cells. Their configuration changes when a photon is absorbed. This triggers a biochemical cascade which can ultimately lead to changes in membrane potential of the receptor cell and thus a signal that is sent to the brain.
Eusocial: "Truly social" — insect societies which have overlapping generations, cooperative brood care, and reproductive labor division (meaning some females — queens — lay eggs, while others — the workers — don't.
Trophallaxis: Direct exchange of food between members of social insects colonies such as bees or ants. One colony member regurgitates food (e.g. nectar) that it has collected elsewhere, and another member immediately picks it up.
Pheromone: chemical substance released by an animal with the intent of influencing another animal's behavior.
Systematics: The science of diversity of animals, founded by Linné - it seeks to classify and organise this diversity into a hierarchy that reflects the degree of relatedness of different animal species.
Lars Chittka is assistant professor of biology at Zoologie II, Biozentrum, Am Hubland, 97074 Würzburg, Germany. He studied biology at the Universities of Goettingen and Berlin. He received his PhD in R. Menzel's lab in Berlin, working on the psychophysics and evolutionary ecology of bee color vision. He then moved to Stony Brook, USA to do a postdoc on the behavioral ecology of bumble bees, working with J.D. Thomson. In Würzburg, his group is exploring bee vision and cognition in the context of evolutionary ecology, and its links with plant pollination and evolution.
Anna Dornhaus is a PhD student in L. Chittka's lab at Zoologie II, Biozentrum, Am Hubland, 97074 Würzburg, Germany. She studied biology and computer science in Freiburg and Würzburg, and recently completed her master's thesis during which she described the previously unknown bumble bee communication system. She is also interested in computer modeling of insect societies and task allocation.