

OCEANOGRAFIA |
Phosphorus in the Upper Ocean |
 Acroread
Acroread
|
© Claudia Benitez-Nelson, 2000
cbnelson@soest.hawaii.edu, Universidad de Hawaii en Manoa, Honolulu, Hawaii, EE.UU. |
[RESUMEN] ABSTRACT
Phosphorus (P) is a key element in the growth of microorganisms throughout the world’s oceans. However, little is known about the cycling of this nutrient within coastal and open ocean environments. In general, phytoplankton consume P for their nutrient needs in the form of HPO42-. As a result, scientists have often ignored the other P containing pools. Recent evidence, however, suggests that bacteria may produce an additional source of inorganic P by remineralizing specific P containing organic compounds. Thus, the importance of other P pools can no longer be overlooked. An overview of upper ocean P cycling is given in the following discussion and includes the latest information regarding the chemical composition, bioavailability, and utilization of P within marine ecosystems.
Phosphorus is an essential nutrient utilized by all living organisms. Yet little is known about the role it plays in limiting the growth and distribution of marine phytoplankton (Fig. 1). These microscopic organisms are at the base of the foodweb. Thus, their abundance is a major controlling factor in foodweb composition throughout the oceanic realm. In addition, phytoplankton consume CO2, a greenhouse gas, during photosynthesis and convert the carbon into biomass during growth. During phytoplankton grazing and egestion, zooplankton produce particles that sink below the upper layers of the ocean. Phytoplankton will also sink from the ocean’s surface via aggregation into larger particles, especially during blooms. In this manner, CO2, in the form of sinking particulate organic matter, is removed from the upper ocean and prevented from interacting with the atmosphere on timescales of 1000 years (Fig. 2). In fact, the oceans may be responsible for the removal of up to 50% of the total anthropogenic CO2 emitted to the atmosphere over the last century (Quay et al., 1992). Sinking organic material also removes pollutants, such as lead, which adsorb to the particles’ surfaces as they sink through the water column (Wageman & Muir, 1994). By this process, these harmful contaminants are removed from the upper ocean, thereby limiting their potential uptake and bioaccumulation by marine animals, which may eventually be eaten by humans.
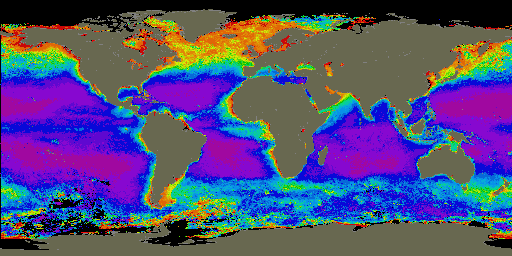

Figure 1. Global distribution of chlorophyll (phytoplankton) from November 1978 to June 1986 as seen by satellites. Red is equivalent to higher phytoplankton concentrations and purple is equivalent to low phytoplankton concentrations. Image was used courtesy of the SeaWiFS Project Home Page (http://seawifs.gsfc.nasa.gov/SEAWIFS.html).)
So what controls the distribution and growth of phytoplankton in the upper ocean? Physical (e.g., light, temperature, mixed layer depth), chemical (e.g., nutrient concentrations) and biological (e.g., grazing) factors may control phytoplankton growth. The role of phosphorus (P) in nutrient limitation depends on the accessibility of other nutrients and trace elements, such as iron. The availability of these nutrients may in turn vary seasonally and spatially. Unfortunately, understanding which nutrient is limiting growth and when, is complicated. It is necessary to know the nutrient concentration, the form in which it is consumed, and its recycling rate. The relative importance of each of these factors is highly contested within the scientific community (Hecky & Kilham, 1988; Codispoti, 1989; Hutchins & Bruland, 1998). Nonetheless, P has been observed to limit phytoplankton production in regimes ranging from restricted, shallow-marine areas (Krom et al., 1991; Fourqurean et al., 1992) to the open ocean, oligotrophic sites of the North Atlantic and North Pacific (Cotner et al., 1997; Karl et al., 1997).
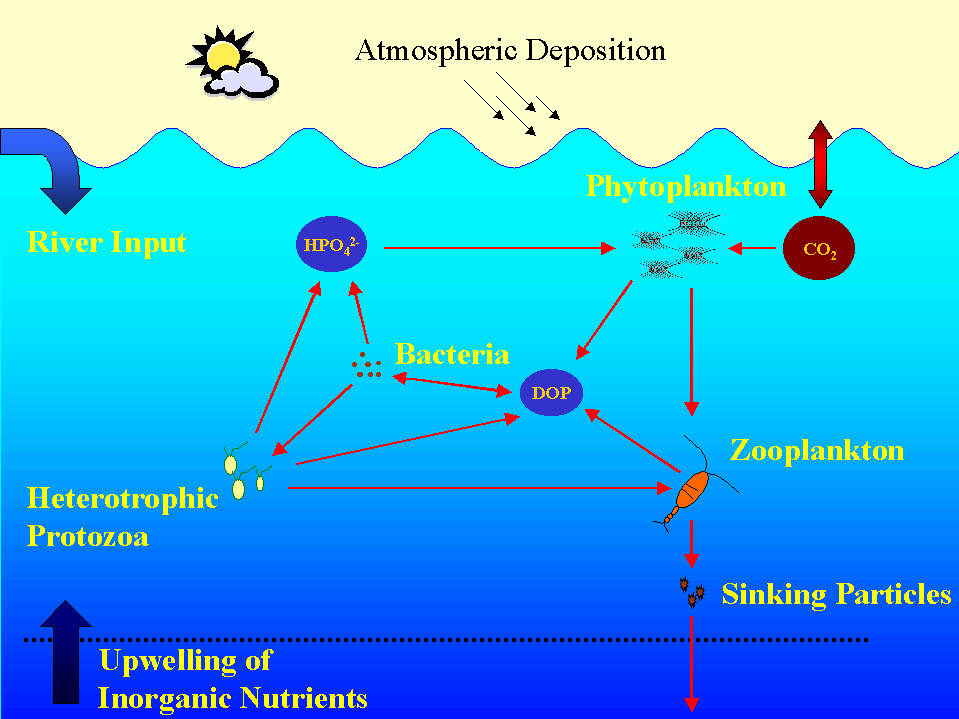
Figure 2. The upper ocean P cycle.
Even in instances when P is not a limiting factor, increasing anthropogenic P inputs in coastal ecosystems may substantially change the dynamics within these fragile communities. In the heavily populated areas of the world’s coastlines, the anthropogenic inputs of P into the coastal ocean (e.g. from fertilizers in agriculture and detergents) are 10-100 times greater than in pre-industrial times (Caraco, 1993). The consequences of such an increase are numerous and include reducing food web diversity, altering phytoplankton compositions and an increase in the intensity and frequency of red tides (e.g. Nixon, 1993). These transformations could also greatly effect phytoplankton growth and zooplankton grazing, thereby potentially altering the extent of particulate matter production in the coastal ocean. While coastal zones represent less than one tenth of the area of the world’s oceans, they are responsible for 20-40% of the marine particulate matter that is exported from the ocean’s surface (Eppley & Peterson, 1978). Thus, it is essential that we understand the role that P plays in these environments.
Phosphorus sources, composition, and sinks
P enters the coastal oceans predominantly through rivers. Estimates of the dissolved inorganic P input range from 3-15 x 1010 mol P yr (Delaney, 1998). Atmospheric deposition of P which is soluble in seawater, has been estimated to be significantly lower, between 1-2 x 1010 mol P yr-1 (Duce, 1986). Although small, the atmospheric deposition of P has been increasingly recognized as being important in the open ocean. In the upper water column, inorganic P (HPO42-) is depleted via phytoplankton growth and incorporation into sinking organic matter. In general, this particulate material is remineralized at depth, such that typical profiles show large depth gradients in P concentrations (Fig. 3). P is subsequently brought back into the surface ocean via physical mixing (upwelling and diffusion). These processes are responsible for some of the most productive fishing areas in the world, such as off the coast of Peru.
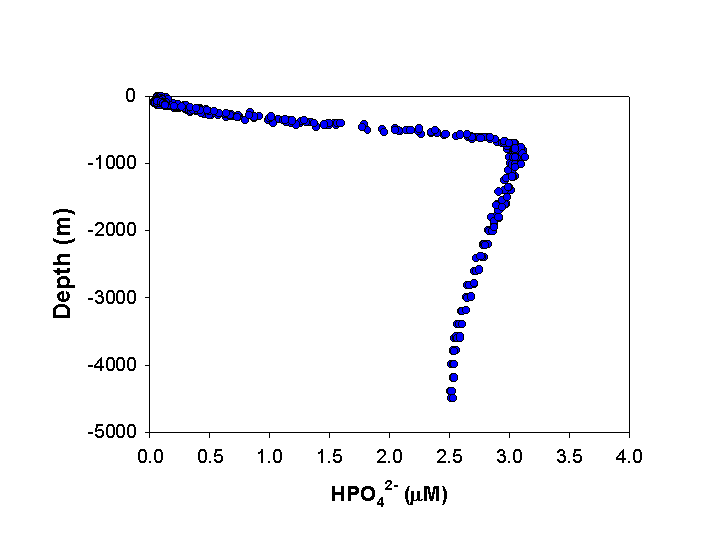
Figure 3. General distribution of inorganic P (SRP) with depth in the open ocean. Example below was data taken from October 1988 to February 1989 at Station ALOHA in the central North Pacific, as part of the Hawaiian Ocean Time-series program (http://hahana.soest.hawaii.edu/hot/hot-dogs/interface.html).
Currently, the distribution of P in aquatic systems is defined analytically. The dissolved P pool is generally defined as the material that passes through a 0.2 - 0.7 micron diameter filter. In most studies, the dissolved inorganic P pool (presumably as HPO42-) is characterized as the fraction which reacts under acidic conditions (pH < 2) to produce a colored compound, phosphomolybdate (Murphy & Riley, 1962). A number of investigations, however, have found that this treatment will also include an unknown proportion of acid-labile organic compounds, such as sugar phosphates (McKelvie et al., 1995, and references therein). A more appropriate term for the above fraction is soluble reactive phosphorus or SRP. The concentration of total dissolved P (TDP) is generally quantified by using high temperature and/or high pressure in the presence of a strong oxidizing reagent (McKelvie et al., 1995). In this manner, all of the P within a sample is converted to inorganic P and measured using the above phosphomolybdate method. The difference between TDP and SRP is then often referred to as the dissolved organic P (DOP) component. This DOP fraction, however, can also contain non-reactive inorganic compounds, such as polyphosphates. Thus, this fraction should really be termed soluble non-reactive P or SNP.
In coastal marine environments, the SRP pool ranges from 50-100% of the TDP pool (Table 1). In contrast, in the open ocean, SRP is often less than 25% (Karl & Yanagi, 1997). As a result, the importance of identifying the composition of the SNP pool increases with greater distance from shore. Employing a variety of techniques, several classes of SNP compounds have been shown to exist in seawater and include polyphosphates, nucleotides, nucleic acids, phosphonates and monophosphate esters (Solerzano & Strickland, 1968; Karl & Bailiff, 1989; Nawrocki & Karl, 1989; Clark et al., 1998). The actual percent composition of SNP, however, has remained elusive. Recently, Karl and Yanagi (1997) utilized more sensitive techniques to characterize part of the SNP pool within the oligotrophic North Pacific. In the upper water column (<100 m), they found that 66% of the SNP pool was most likely dominated by monophosphate esters, compounds used by phytoplankton for catabolic functions. In contrast, in the deeper waters this fraction decreased to less than 50% of the SNP Pool, indicating preferential remineralization of one component over the other.
Table 1. General distribution and composition of dissolved P in the oceans. See text for definitions.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional evidence for a more ‘bioavailable pool’ was found using 31P Nuclear Magnetic Resonance (NMR). In this study, Clark and coworkers (1998) found that the high molecular weight fraction of dissolved organic P (1 -100 nm size range), or 19-38% of the total dissolved organic matter, was dominated by phosphorus esters and phosphonates. In plankton phosphonates are associated with phosphonolipids, compounds used in cell structure. Their abundance within marine phytoplankton, however, was almost ten times lower than that found in the dissolved organic phosphorus pool. This result again suggests that other P containing components must be preferentially removed in order to increase the relative concentration of phosphonates in the water column.
The above studies have provided indirect evidence that there is a pool of P within marine systems which is being preferentially eliminated from the water column. How does this removal occur? It is most likely by marine bacteria (Azam, 1998). Bacteria contain enzymes located within the outer membrane that are capable of liberating inorganic P from organic compounds. Alkaline phosphatase and 5’ nucleotidase are two of the major enzyme classes found in marine bacteria (Ammerman & Azam, 1985). Over the last several years, laboratory studies have demonstrated that bacteria readily degrade compounds such as monophosphate esters and nucleotides, through the use of these enzymes (Ammerman & Azam, 1985; Bjorkman & Karl, 1994). In fact, using diagnostic enzymatic assays, between 10-50% of the SNP in coastal marine environments could be available (Strickland & Solorzano, 1966; Taft et al., 1977; Kobori & Taga, 1979). The role of bacteria in the cycling of organic matter in the open ocean, where the concentration of SNP relative to TDP is greater, may be even more significant.
Bacteria are the major food source for heterotrophic protozoa. Laboratory studies have demonstrated that through feeding and egestion, protozoa can convert between 20-90% of the P within the bacterial biomass to soluble forms (Anderson et al., 1986; Jurgens & Gude, 1990; Ferrier-Pages & Rassoulzadegan, 1994; Eccleston-Parry & Leadbeater, 1995). The dissolved P produced from these processes is dominated by inorganic P, although organic P is also formed (Anderson et al, 1986; Jurgens & Gude, 1990). The efficiency of P regeneration appears to be dependent on the nutrient-limitation conditions, as well as the phase of protozoan growth (Anderson et al., 1986; Jurgens & Gude, 1990). Controversy still exists over the specific role that protozoans play in nutrient regeneration in natural systems. In fact, there is little direct evidence of the magnitude of bacterial and heterotrophic protozoa remineralization processes and their effect on phytoplankton production in general. In order to constrain their importance, we must also identify the P recycling rates within each of these biological pools.
Phosphorus Recycling Rates
One method of elucidating the timescales over which nutrient recycling occurs is through the use of radioisotopes. In essence, radioisotopes behave as a ‘clock’, enabling one to trace processes that have occurred over a well-defined time interval. There are three isotopes of P, one stable, 31P, and two radioactive, 32P (t_ = 14.3 days) and 33P (t_ = 25.3 days). Using 32P and 33P one can monitor the movement of P throughout various biological reservoirs and determine a net ‘age’ of P within each particular pool. More simply, the ratio of 33P/32P will increase with time due to the differential decay rates of 32P and 33P. These tracers have been used to investigate the recycling rate of P within a number of aquatic regimes. In the past, the use of these isotopes was restricted to incubation experiments in which artificially produced 32P and 33P had been added. The difficulty with such types of research is that they involve significant perturbations to the system of interest. For example, samples are first separated from the ecosystem before incubation. Bottle incubations will, at best, miss sporadic bloom events and provide rate estimates that are only valid for discrete depths and times. Nonetheless, these studies have found that phytoplankton and bacteria rapidly consume P and that there was rapid exchange between inorganic and organic P pools (Perry & Eppley, 1981; Orret & Karl, 1987; Bjorkman & Karl, 1994).
How does this relate to the oceanographic realm? Over the last decade, several researchers have used naturally produced 32P and 33P to directly investigate the cycling of marine P. 32P and 33P are naturally produced in the atmosphere via cosmic ray interactions with atmospheric argon nuclei. Once produced, these isotopes quickly scavenge onto particles and enter the oceans predominantly in rain. While the magnitude of 32P and 33P concentrations will vary dramatically from one rain event to another, studies have shown that in general, the ratio of 33P/32P remains constant (Waser & Bacon, 1995; Benitez-Nelson & Buesseler, 1999a). The advantage of using naturally produced 32P and 33P is that they enable scientists to examine the recycling of P without disturbing the regime of interest. Furthermore, results will integrate over all of the processes that have affected the distribution of 32P and 33P over the prior 20 to 35 days. Preliminary results provided evidence that this type of investigation has enormous potential for understanding P cycling in marine systems (Lal & Lee, 1988; Waser et al., 1996). In general, it was found that the age of P increased as one moved up through the food chain. In the open ocean, the recycling rate of P in zooplankton ranged from 60-70 days (Waser et al., 1996). In contrast, the recycling rate of zooplankton off the coast of California was less than 40 days (Lal & Lee, 1988). Measurement of 32P and 33P in the SRP and SNP pools also indicated that the recycling time of SRP was short, while that of SNP was substantially longer at 6 weeks (Lal & Lee, 1988). Unfortunately, these results had large uncertainties due to the fact that naturally produced 32P and 33P were very difficult to measure in the field.
New advances in technology have enabled a much more comprehensive investigation of P recycling using 32P and 33P. Within the Gulf of Maine, 32P and 33P were measured in SRP, SNP and TDP as well as in various biological pools, including bacteria, throughout the Spring and Summer months (Benitez-Nelson & Buesseler, 1999b). In this study, it was found that dissolved P recycling rates varied significantly from season to season. SRP turnover rates were always high, regardless of the actual concentration. SNP turnover rates, on the other hand, ranged from a month to greater than three months. In the summer, marine bacteria were consuming SNP directly, even though there were measurable amounts of SRP. While the bacterially-consumed SNP was only a small fraction of the total SNP, it is highly likely that this material was younger than the rest of the SNP pool.
What does this all mean in terms in of the cycling of P? The rapid cycling of SRP indicates that low P concentrations can support much higher levels of biological production than previously thought. In contrast, the higher turnover rates of SNP imply that much of this pool is not available for biological uptake. Nonetheless, there is a small fraction of SNP that can be directly utilized by bacteria, indicating that SNP in this regime is made up of a chemically diverse group of compounds that cycle over very different timescales. The observation that bacteria were consuming SNP in the presence of SRP, suggests that bacteria were remineralizing SNP for other nutrient requirements, such as for carbon or nitrogen. Regardless of why, the evidence that bacteria are consuming specific organic P compounds within marine ecosystems is novel and indicates that this process can provide a new source of inorganic nutrients to phytoplankton. Interestingly, there is no evidence that heterotrophic protozoa consume bacteria, or are significantly involved in the P remineralization process.
This study (Benitez-Nelson & Buesseler, 1999b) provided the first direct evidence that marine bacteria may preferentially remineralize specific compounds of SNP. Since this bacterially-utilized SNP fraction was younger than the bulk DOP pool, it most likely consists of P compounds that have rapid recycling rates within marine plankton. These include such compounds as monophosphate esters and nucleotides, which have been shown to be preferentially remineralized in the laboratory. Thus, we now have a clearer picture of the types of P compounds that are available for bacterially-mediated remineralization. Furthermore, 32P and 33P represent an effective technique to further investigate the cycling of P in the oceans.
Conclusions
Changes within the biogeochemical cycle of P have the potential to impact the world in many different ways. Thus, it is essential that we understand the distribution, composition, and recycling rate of P within the oceanic realm. Knowledge of the composition of P within marine systems, however, remains lacking. In order to fully describe the P composition new methods need to be developed. The indication that bacteria are remineralizing specific organic P compounds greatly impacts how scientists currently view nutrient limitation. This information, along with recent studies that demonstrate rapid recycling of SRP, will need to be included in all future models of phytoplankton distributions and their role in the uptake of CO2 and other anthropogenic components.
References
Ammerman, J. W. & Azam, F. (1985) Bacterial 5’-nucleotidase in aquatic ecosystems: A novel mechanism of phosphorus regeneration. Science, 227, 1338-1340.
Anderson, O. K., Goldman, J. C., Caron, D. A. & Dennett, M. R. (1986) Nutrient cycling in a microflagellate food chain: III. Phosphorus dynamics. Mar. Ecol. Prog. Ser., 31, 47-55.
Azam (1998) Microbial control of oceanic carbon flux: The plot thickens. Science, 280, 694-696.
>Benitez-Nelson, C.R. & Buesseler, K. O. (1999a) Phosphorus 32, phosphorus 33, beryllium 7, and lead 210: Atmospheric fluxes and utility in tracing stratosphere/troposphere exchange. J. Geophys. Res., 104, 11,745-11,754.
Benitez-Nelson, C.R. & Buesseler, K. O. (1999b) Temporal variability of inorganic and organic phosphorus turnover rates in the coastal ocean. Nature, 398, 502-505.
Bjorkman, K. & Karl, D. M. (1994) Bioavailability of inorganic and organic phosphorus compounds to natural assemblages of microorganisms in Hawaiian coastal waters. Mar. Ecol. Prog. Ser., 111, 265-273.
Caraco, N. F. (1993) Disturbance of the marine phosphorus cycle: a case of indirect effects of human activity. Trends Ecol. Evol., 8/2, 51-54.
Cho, B. C. & Azam, F. (1988) Major role of bacteria in biogeochemical fluxes in the ocean’s interior. Nature 332, 441-443.
Clark, L. L., Ingall, E. D. & Benner, R. (1998) Marine phosphorus is selectively remineralized. Nature 393, 426.
Codispoti, L.A. (1989) Phosphorus vs. Nitrogen Limitation of New and Export Production. In: Productivity of the Ocean: Present and Past, W. H. Berger, V. S. Smetacek, G. Wefer (Eds.), John Wiley & Sons Limited, 377-394.
Cotner, J. B., Ammerman, J. W., Peele, E. R., & Bentzen, E. (1997) Phosphorus-limited bacterioplankton growth in the Sargasso Sea. Aquatic Microb. Ecol., 13, 141-149.
Delaney, M. L. (1998) Phosphorus accumulation in marine sediments and the oceanic phosphorus cycle. Global Biogeochem. Cycles, 12, 562-572.
Duce, R. A. (1986) The input of atmospheric nitrogen, phosphorus, and iron species on marine biological productivity. In The Role of Air-Sea Exchange in Geochemical Cycling, P. Buat-Menard (Ed.), D. Reidael Publishing Company, Dordrecht, The Netherlands, 497-529.
Eccleston-Parry, J. & Leadbeater, B. S. (1995) Regeneration of phosphorus and nitrogen by four species of heterotrophic nanoflagellates feeding on three nutritional states of a single bacterial strain. Appl. and Environ. Microb., 61, 1033-1038.
Eppley, R. W. & Peterson, B. J. (1978) Particulate organic matter flux and planktonic new production in the deep ocean. Nature, 282, 677-680.
Ferrier-Pages, C. & Rassoulzadegan, F. (1994) Remineralization in planktonic protozoa. Limnol. Oceanogr., 39, 411-419.
Fourqurean, J. W., Ziemen, J. C., & Powell, V. N. (1992) Phosphorus limitation of primary production in Florida Bay: Evidence from C:N:P ratios of the dominant seagrass Thalassia testudinum. Limnol. Oceanogr., 37, 162-171.
Hecky, R. E. & Kilham, P. (1988) Nutrient limitation of phytoplankton in freshwater and marine environments: A review of recent evidence on the effects of enrichment. Limnol. Oceanogr., 33, 796-822.
Hutchins, D. A. & Bruland, K. W. (1998) Iron-limited diatom growth and Si:N ratios in a coastal upwelling regime. Nature, 393, 561-564.
Jurgens, K & Gude, H. (1990) Incorporation and release of phosphorus by planktonic bacteria and phagotrophic flagellates. Mar. Ecol. Prog. Ser., 59, 271-284.
Karl. D. M. & Baliff, M. D. (1989) The measurement and distribution of dissolved nucleic acids in aquatic environments. Limnol. Oceanogr., 34, 543-558.
Karl, D. M. & Yanagi, K. (1997) Partial characterization of the dissolved organic phosphorus pool in the oligotrophic North Pacific Ocean. Limnol. Oceanogr. 42, 1398-1405.
Kobori, H. & Taga, N. (1979) Phosphatase activity and it’s role in the mineralization of organic phosphorus in coastal seawater. J. exp. Mar. Biol. Ecol., 36, 23-39.
Krom, M. D. Kress, N., Benner, S., & Gordon, L. I. (1991) Phosphorus limitation of primary productivity in the eastern Mediterranean Sea. Limnol. Oceanogr., 36, 424-432.
Lal, D. & Lee, T. (1988) Cosmogenic 32P and 33P Used as Tracers to Study Phosphorus Recycling in the Upper Ocean. Nature, 333, 752-754.
McKelvie, I. D., Peat, D. M., & P. J. Worsfold (1995) Techniques for the quantification and speciation of phosphorus in natural waters. Anal Proc. Including Anal. Comm., 32, 437-445.
Murphy, J. & Riley, J. P. (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim. Acta, 27, 31-36.
Nawrocki, M. P. & Karl, D. M. (1989) Dissolved ATP turnover in the Bransfield Strait, Antarctica during the spring bloom. Mar. Ecol. Prog. Ser., 57, 35-44.
Nixon, S. W. (1993) Nutrients and coastal waters: too much of a good thing? Oceanus, 36/2, 38-47.
Orrett, K & Karl, D. (1987) Dissolved organic phosphorus production in surface waters. Limnol. Oceanogr., 32, 383-395.
Perry, M. J. & R. W. Eppley (1981) Phosphate uptake by phytoplankton in the central North Pacific Ocean. Deep-Sea Research, 28, 39-49
Quay, P. D., Tilbrook, B. & Wong, C. S. (1992) Oceanic uptake of fossil fuel CO2: Carbon-13 evidence. Science, 256, 74-78.
Strickland, J. D. H. & Solorzano, L (1966) Determination of monoesterase hydrolysable phosphate and phosphomonoesterase activity in sea water. In: Some contemporary studies in marine science, H. Barnes (Eds.), G. Allen & Unwin Ltd., London, pp. 665-674.
Solorzano, L. & Strickland, J. D. H. (1968) Polyphosphate in seawater. Limnol. Oceanogr., 13, 515-518.
Taft, J. L., Loftus. M. E. & Taylor, W. R. (1977) Phosphate uptake from phosphomonoesters by phytoplankton in the Chesapeake Bay. Limnol Oceanogr., 22, 1012-1021.
Wageman, R. & Muir, D. C. G. (1994) Concentrations of heavy metals and organochlorides in marine mammals of northern waters: Overview and evaluation. Canadian Technical Report Fisheries Aquatic Science, No. 1279, pp. 97.
Waser, N. A. D. & Bacon. M. P (1995) Wet deposition fluxes of cosmogenic 32P and 33P and variations in the 33P/32P ratios at Bermuda. Earth Planet. Sci. Lett., 133, 71-80.
Waser, N. A. D., Bacon. M. P. & Michaels, A. F. (1996) Natural activities of 32P and 33P and the 33P/32P ratio in suspended particulate matter and plankton in the Sargasso Sea. Deep-Sea Res. 43(2-3), 421-436.
WWW sites of Interest
Educational information on the sea, very informative!
http://www.seasky.org/sea.html
Look at ocean color (phytoplankton distributions) using satellites.
http://seawifs.gsfc.nasa.gov/seawifs/living_ocean/living_ocean.html
Learn more about algal blooms and Red Tides
http://www.redtide.whoi.edu/hab/
See neat pictures of marine phytoplankton
Glossary
Bacteria: Ubiquitous in nature, these prokaryotic organisms lacks organelles, such as nuclei.
Bioaccumulation: The process by which marine animals consume harmful components and concentrate them within their tissues.
Blooms: Most common in the spring, these refer to a large increase in phytoplankton abundance at the ocean’s surface.
Cosmic rays: High-energy particles, such as protons and neutrons, which continuously bombard the Earth’s surface.
Greenhouse Gas: One of many gases that are emitted to the atmosphere through man’s activities and has been hypothesized to create a global increase in temperature.
Heterotrophic protozoa: Passively floating unicellular organisms that consume organic material.
1 Mol = 6.02 x 1023 molecules.
Nutrient Limitation: In biological systems, one nutrient tends to be depleted first during growth, and must be re-supplied for further growth to occur.
Oligotrophic: Those aquatic systems that are low in the nutrients which support the growth of phytoplankton.
Phytoplankton: Passively floating photosynthetic organisms which dominate the marine environment and include the three domains of life: Bacteria, Archea, and Eukarya.
Radioisotopes: Elements that are unstable, such that they decay to a more stable form via the emission of radiation. Radioisotopes are characterized by half-lives (t_), which is the length of time necessary for exactly half of the number of original radionuclide atoms to decay away. For example, after two half-lives have passed, only one quarter of the starting radionuclide material will still exist. By using the difference between the known initial radioisotope concentration and what is actually measured in the sample, one can estimate how much time has elapsed since sample formation. This is the basis for many radioisotope-dating techniques.
Red Tides: An aquatic phenomena caused by blooms of toxin-producing dinoflagellates. Concentrations of these species are high enough to give the water a red-brown appearance.
Remineralization: The process by which organic material is converted back into simpler, inorganic forms.
Zooplankton: Passively floating animals that consume phytoplankton.